Stryker has introduced the MAKO Robotic Power System (RPS), a handheld addition to its broader MAKO portfolio. The device received FDA clearance in August 2025 for total knee arthroplasty (TKA) and is currently undergoing a limited market release. A wider showcase is expected at the American Academy of Orthopaedic Surgeons annual meeting in March 2026.
The launch adds another layer to the competitive landscape in orthopedic surgical robots, particularly in knee replacement. Before evaluating the strategic implications, it is worth revisiting the market fundamentals driving this category.
The Numbers Behind Robotic Knee Surgery
TKA continues to grow at a meaningful pace in the U.S. Approximately 1.6 million TKAs were performed in 2025. LSI projects that knee replacement volumes will outpace overall orthopedic procedure growth, expanding at a compound annual growth rate of 8.4% between 2025 and 2030. By 2030, annual procedure volume is expected to reach roughly 2.4 million.
Robotic assistance now accounts for a significant portion of these procedures. LSI estimates that about 25% of U.S. TKAs are performed robotically. This figure reflects triangulated inputs from multiple companies across the market, including:
- THINK Surgical: ~23%
- Stryker: 60% of all Stryker knee implants
- Johnson & Johnson: 25% of all J&J knee implants
- Zimmer Biomet: 20%–25%
Applied to overall volume, that equates to roughly 400,000 knee replacements performed annually with a surgical robot.
The market has shown strong demand for robotic assistance in knee replacement procedures. This is backed by strong clinical data showing that knee replacement surgery benefits from reduced rates of implant malposition, perioperative fracture, and early revision compared to manual knee replacement surgery. The data also suggests that patients benefit from improved early pain and function scores. Adoption is being driven by both clinical outcomes and provider and patient preferences.
The trajectory is clear. Robotic knee surgery is no longer niche. It is becoming foundational, which makes Stryker’s latest move particularly notable.
Stryker and THINK Surgical
Stryker already holds more than 75% market share in the U.S. for orthopedic surgical robots and has installed more than 1,500 systems in the U.S. On paper, that is a commanding position.
Yet the MAKO RPS enters a space that has recently been validated by a different approach to knee robotics.
THINK Surgical’s TMINI Miniature Robotic System, which was first cleared in 2023, is a wireless handheld robotic handpiece for total knee replacement. The design focuses on flexibility for both surgeons and facilities. Rather than requiring the traditional footprint of a large robotic system, TMINI was built to accommodate environments where space and workflow efficiency matter.
During LSI USA 2025, CEO Stuart Simpson shared updates on THINK Surgical’s positioning and commercial momentum:
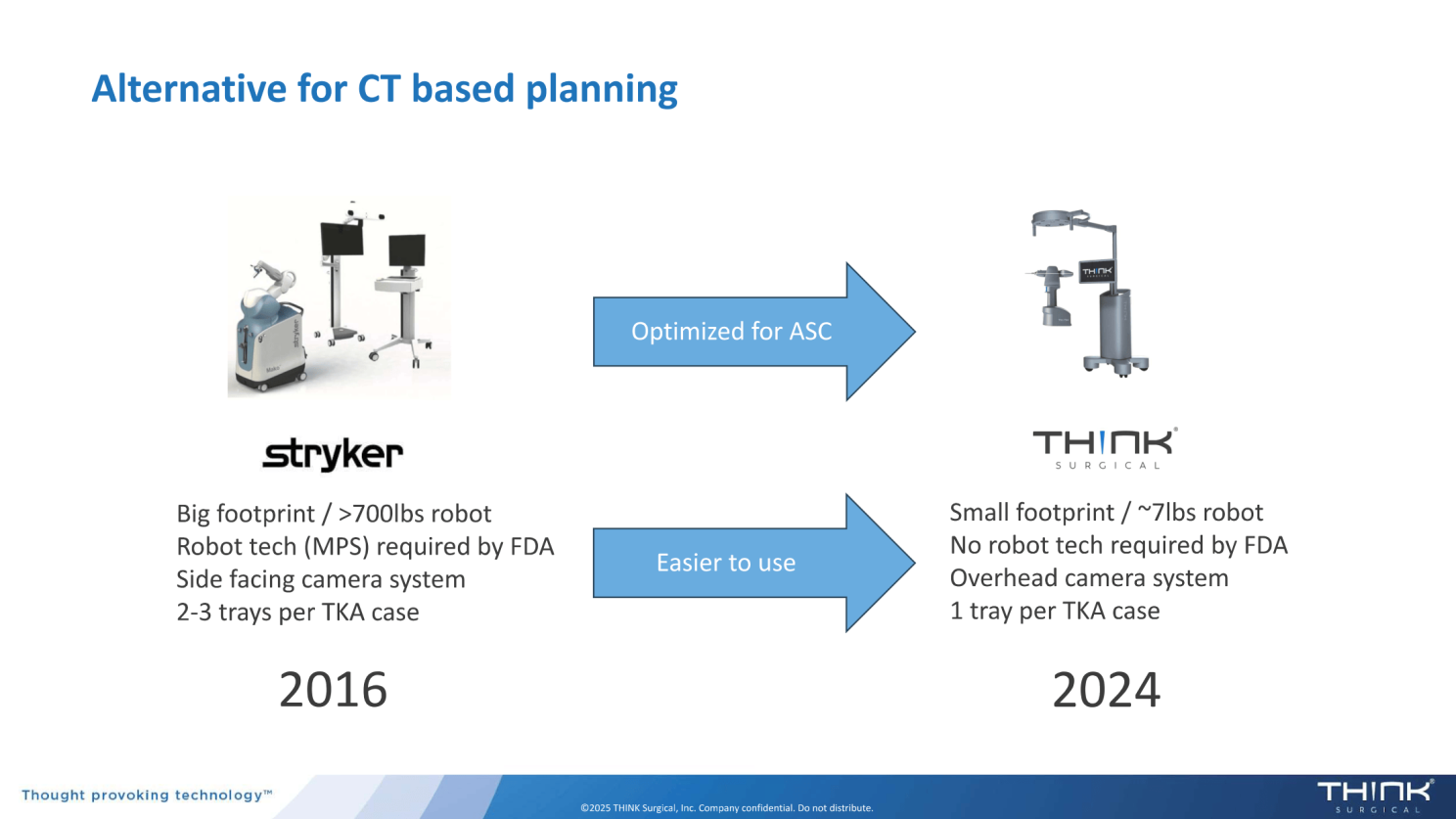
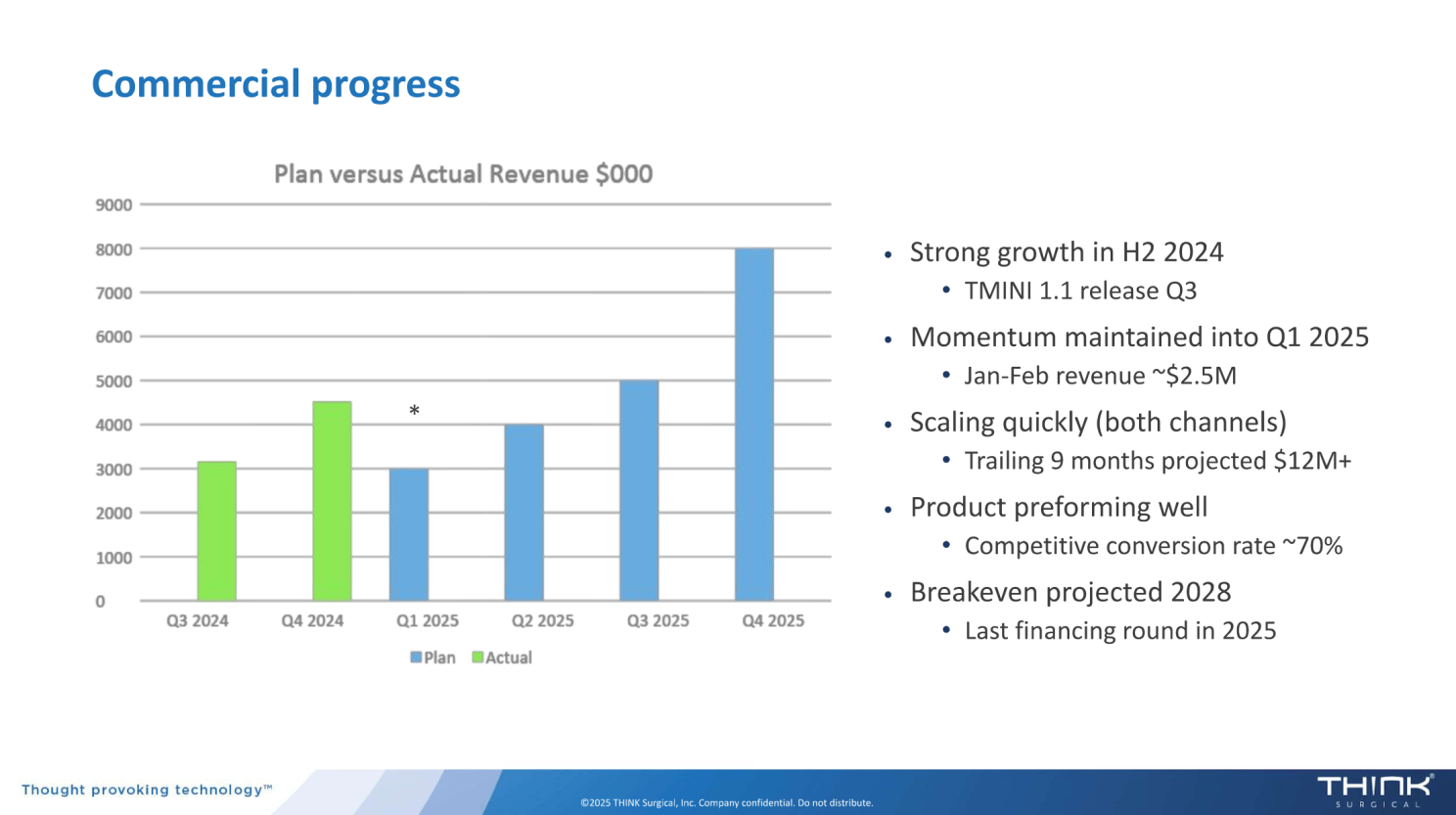
Against this backdrop, MAKO RPS appears strategically aligned. A handheld robotic power system allows Stryker to reach similar ASC-focused customers and reinforce its leadership position before meaningful share shifts occur.
To gain a deeper understanding of THINK Surgical’s growth and Stuart Simpson’s perspective on the competitive landscape, access Compass AI today.
The Shift Toward Smaller Orthopedic Surgical Robots
Across surgical robotics, a consistent theme is emerging. Systems are becoming more compact and better suited for outpatient environments. As procedures migrate into ambulatory settings, large footprints and complex infrastructure requirements become limiting factors.
Control of the robotic approach brings broader strategic advantages. Robotics platforms influence implant selection, consumables, and data capture. In practical terms, whoever owns the robot shapes the downstream economics.
The introduction of MAKO RPS is more than a product update. It reflects recognition that miniaturization and workflow alignment are becoming competitive necessities.
The question is no longer whether handheld robotic systems have a role in knee surgery. The question is, who else will follow?
Stryker at LSI USA ‘26
Stryker leaders Adam Wollowick and Aaron Johnson will take the stage at LSI USA ‘26 to discuss how to identify an investment, what they want to see before investing and acquiring, and what actually gets deals done in today’s market.
Looking Ahead
The introduction of MAKO RPS reinforces where the market is heading. As procedures continue to move into outpatient environments and competition intensifies, the next chapter of orthopedic surgical robots will be shaped by portability, workflow efficiency, and the ability to meet surgeons where they operate.
17011 Beach Blvd, Suite 500 Huntington Beach, CA 92647
714-847-3540© 2026 Life Science Intelligence, Inc., All Rights Reserved. | Privacy Policy | Your Privacy Choices | Delete my Data