Under the direction of CEO Ken Mariash, Scientific Co-Founders Emiliano Santarnecchi, PhD, and Giacomo Koch, MD, PhD, and CCSO Lisa Fosdick, Sinaptica Therapeutics is advancing a personalized, non-invasive neuromodulation platform designed to treat Alzheimer’s disease by directly targeting dysfunctional brain networks. Built on years of foundational research and supported by positive Phase 2 data, the company is developing a precision therapy that pairs transcranial magnetic stimulation (TMS) with EEG-based brain mapping, machine learning, and neuronavigation to deliver patient-specific treatment. With a growing body of published data, an established partnership with Nexstim, new Category III reimbursement codes, and plans to enroll patients in a seamless adaptive pivotal trial in late 2026, Sinaptica is working to bring a new therapeutic model to one of medicine’s largest unmet needs.
Origin Story
Sinaptica was founded to translate over a decade of neuroscience research into a scalable therapy for Alzheimer’s disease. Scientific co-founders Dr. Emiliano Santarnecchi and Dr. Giacomo Koch had spent years studying how neurodegenerative diseases disrupt brain networks, while future president Rich Macary was drawn to the space after his mother was diagnosed with Alzheimer’s and he began searching for better treatment options. Their work came together around a core idea that still defines the company today. Alzheimer’s is not just a disease of plaques and proteins; it is a disorder of large-scale brain networks, particularly the Default Mode Network (DMN), which plays a central role in memory and identity.
That insight helped shape a Phase 2 clinical trial in mild-to-moderate Alzheimer’s patients at the Santa Lucia Foundation in Rome, where targeted TMS combined with EEG-based brain mapping generated promising results. Sinaptica was then built as a clinical company to commercialize that work, launching with positive Phase 2 data, proprietary targeting software integrating TMS with EEG and neuronavigation, hardware manufacturing capabilities, and a strong IP portfolio.
For Santarnecchi, the opportunity was compelling from the start because it offered a way to move from lab-based insight to real patient impact. “I was excited to co-found Sinaptica Therapeutics because it gave us the opportunity to translate years of research on brain connectivity and neuroplasticity into a therapy that could meaningfully impact Alzheimer’s patients,” Santarnecchi said. “The idea of a non-drug, non-invasive approach that directly addresses a true unmet need was incredibly motivating.”
Mariash later joined the company after a career that included senior roles within Boston Scientific’s Neuromodulation division, where he led global strategy and portfolio management, contributing to business development. What drew him in was both the magnitude of the clinical problem and the distinctiveness of Sinaptica’s approach. “When I first engaged with Sinaptica, what stood out was the rigor of the science paired with a new approach to one of the largest unmet needs in medicine,” Mariash said. “The science is compelling, the clinical data are consistent and strong, and the potential to reach a broad population of patients without systemic side effects is incredibly meaningful.”
The Current Landscape
The need for new Alzheimer’s therapies continues to grow. Sinaptica notes that about 8.5 million Americans are projected to be affected by Alzheimer’s disease by 2030, rising to 13.8 million by 2060. At the same time, current treatment options remain limited. Existing FDA-approved therapies fall into two categories: symptomatic treatments, such as cholinesterase inhibitors and memantine-based drugs, and anti-amyloid monoclonal antibodies intended to slow decline in carefully selected early-stage patients with confirmed amyloid pathology. The former do not appear to alter disease course, while the latter offer only modest slowing of decline and come with meaningful risks, including black box warnings for amyloid-related imaging abnormalities (ARIA).
Sinaptica’s leadership believes part of the problem lies in how Alzheimer’s has traditionally been framed. Rather than viewing it as a disease confined to a specific protein target or brain region, the company argues that Alzheimer’s should be understood as a disease of network dysfunction.
“When I say Alzheimer’s is a disease of network dysfunction, I’m talking about a shift from viewing it as one problem in one spot, like plaques in a region, to understanding it as a breakdown in how the brain communicates as an integrated system,” Mariash said. He points specifically to the DMN, a large distributed network involved in episodic memory, introspection, and self-reference. “In Alzheimer’s, what we consistently see is that the network loses coherence: there are excitation and oscillatory imbalances, signaling becomes dysregulated and disrupted, the network disconnects, and the system loses the ability to ‘read’ and ‘write’ data. In other words, it loses plasticity,” he explained.
This framing matters because it opens a whole new landscape of possible modalities that target Alzheimer’s at the network level. “If the core issue is that the network is failing to coordinate, then a therapy that can restore healthier network dynamics in a targeted, measurable way has the potential to affect multiple domains: cognition, daily function, and behavior,” Mariash said. “It also opens the door to personalization, because network dysfunction isn’t identical in every patient.”
Inside the Innovation
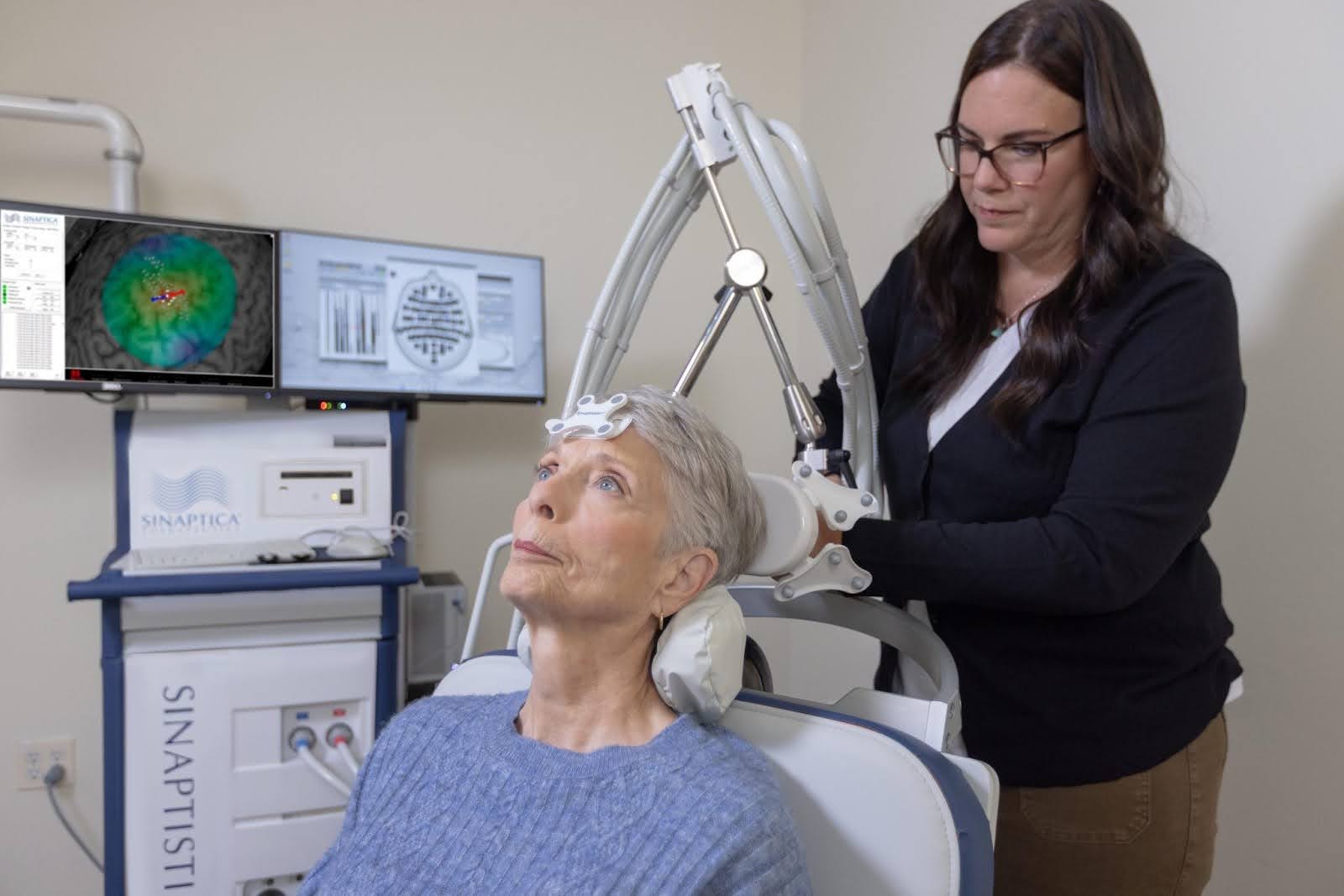
Sinaptica’s therapy is designed to personalize treatment at the network level. By combining TMS with EEG, the company aims to identify how each patient’s brain is wired, determine the optimal stimulation target and dose, and then deliver repeatable therapy with precision over time. The goal is to engage the entire distributed network associated with Alzheimer’s, not just a single location, and to do so in a way that is measurable, adaptable, and reproducible.
“Our focus is on using stimulation to induce neuroplasticity within the DMN, strengthening the connections that are disrupted early in Alzheimer’s,” Koch explained. “Every brain has different network dynamics, so by pairing stimulation with EEG, we can perturb the brain with single pulses, map its connections, and identify the optimal targets for each patient.”
Once customized, the therapy is delivered in weekly 20-minute sessions. Under the hood, the platform is more technically sophisticated than standard TMS workflows. Sinaptica measures the propagation of single pulses of TMS energy in real time using a 64-channel EEG across nine locations on the precuneus in a 3x3 grid. That dataset is processed using machine-learning-derived algorithms that remove artifacts and extract features to identify the right stimulation location, enabling Sinaptica to determine a safe but effective dose unique to the patient. After that calibration step, the company uses neuronavigation to return to the precise target and deliver therapy over time.
Mariash describes TMS-EEG as an EKG of the brain. “Just as an EKG measures the propagation of an action potential through the heart, TMS-EEG measures how single pulses of energy—in our case, delivered to the precuneus—propagate throughout the DMN,” he explained. That gives the company a way not only to personalize therapy but also to objectively measure whether the network is stabilizing over time. “It can also be used to track response to therapy,” Mariash said. “If evoked potentials are not declining over time, it means the network isn’t degrading.”
The company’s proprietary Sinaptistim™ device and MAINTAIN software are central to that vision. Together, they are intended to simplify the translation of complex TMS-EEG data into a clinician-ready treatment report, allowing physicians to deliver a personalized “prescription” without needing to manually clean and interpret EEG data themselves. Sinaptica believes that combination positions the company at the forefront of a new class of precision neuromodulation therapies for neurodegenerative disease.
Progress and Milestones
Sinaptica is a clinical-stage company headquartered in the biotechnology hub in Cambridge, Massachusetts, targeting the U.S. market first while planning selective expansion into the EU and other international markets. The company says it has already begun discussions with a variety of localization partners and prospective customers in Canada, APAC, and the Middle East.
Its near-term roadmap is substantial. “The next 12 months will bring regulatory approvals to use Sinaptistim in clinical trials across North America and the EU,” Fosdick said. “We anticipate the first patients will be enrolled in our seamless adaptive pivotal clinical trial in late 2026. This will be a very important milestone for the company.”
The initial phase of the trial will include five treating centers and a few additional recruitment sites in the U.S. and EU, with a first interim analysis anticipated in 2028. Alongside trial preparation, the company is continuing to build on a strong scientific and operational foundation established by its co-founders and expanded through its internal team.
“We are building on a strong foundation of prior work by our scientific co-founders, which led to strong IP around TMS-EEG. We have FDA Breakthrough Device Designation and are actively engaged in the FDA TAP program, creating a strong regulatory working relationship with the agency,” Macary said. The company has assembled a cross-functional team with experience across TMS development, clinical delivery, and software engineering, including talent from GE, Boston Scientific, Baxter, Sarepta, and Philips. That team is now focused on translating foundational research into scalable product development and multicenter clinical evidence.
Beyond clinical development, Sinaptica has also taken steps toward future commercialization. The company has been granted two new Category III reimbursement codes specific to its calibration and therapy procedures, signaling early recognition of the therapy’s differentiated approach and supporting a pathway toward future coverage.
On the financing side, Mariash noted that Sinaptica has closed an oversubscribed $9 million round, and is now in process on a pivotal round as the company advances toward regulatory approval and commercialization.
For Mariash, the opportunity extends beyond incremental progress to a broader shift in how Alzheimer’s is treated at scale. “Alzheimer’s remains an enormous clinical and economic challenge, and while drug development has made incremental progress, the opportunity to intervene through precision neuromodulation represents a fundamentally different path.”
17011 Beach Blvd, Suite 500 Huntington Beach, CA 92647
714-847-3540© 2026 Life Science Intelligence, Inc., All Rights Reserved. | Privacy Policy | Your Privacy Choices | Delete my Data