Under the direction of Founder and CEO Melissa Krebs, PhD, GelSana is developing a novel class of synthetic polymers designed to address two of the most persistent barriers in chronic wound healing: inflammation and infection. Built on more than a decade of biomaterials research, the company’s technology is engineered to help calm inflammation, repel bacteria, and create an optimal environment for wound healing. With strong preclinical results, nearly $6 million raised to date, and its first product, CleraFlex™ Stretchable Wound Dressing, expected to launch imminently, GelSana is entering a large and rapidly evolving wound care market with a fundamentally new materials platform.
Origin Story
GelSana’s technology originated in the academic lab of Melissa Krebs, PhD, who has spent her career researching biomaterials and translational medical technologies. The polymers that now form the foundation of GelSana were developed and refined in her lab over more than a decade.
“I’ve been doing biomaterials research my whole career,” Krebs explained. “But the products that ultimately became GelSana started in my lab at the Colorado School of Mines more than ten years ago.”
The company itself emerged from an unexpected opportunity. In 2020, Krebs began discussions with a local VC fund in Colorado about the underlying technology. At the time, she was not actively planning to start a company.
“In September 2020, they offered me pre-seed funding to start a company,” she recalled. “That’s when I decided to take the step. I founded GelSana, spun the technology out of my university lab, and licensed the IP from the university into the company.”
The scientific foundation was already well established. Years of research had produced extensive material development data alongside early preclinical validation.
“We had already generated a lot of in vitro data and completed the core material development,” Krebs said. “We were also seeing very promising results in early animal studies showing improved wound healing.”
That body of research gave GelSana a strong starting point, allowing the company to launch with both a differentiated materials platform and encouraging early evidence of clinical potential.
The Current Landscape
Chronic wounds remain one of the most costly, difficult, and often overlooked challenges in healthcare. These wounds, including diabetic foot ulcers, are characterized by persistent inflammation that prevents the body’s natural healing processes from progressing.
Existing wound dressings often focus on protecting the wound or managing moisture. However, they do not directly address the underlying environment that can stall healing in instances where there is too much inflammation, and the wound cannot transition out of this inflammatory phase.
The scale of the problem is enormous. Diabetic ulcers alone are estimated to cost $78 billion annually in the United States. They account for roughly 80% of non-traumatic amputations, and globally, a limb is lost every 20 seconds due to a non-healing diabetic ulcer.
At the same time, the wound care market is undergoing significant change. Krebs noted that shifts in reimbursement policies are creating a window for new technologies.
“The wound care market is notoriously competitive,” she said. “But we’re entering with a completely new material at a time when reimbursement dynamics are shifting significantly.”
Traditionally, many advanced wound therapies have relied on expensive skin substitutes and other cellular and tissue-based products. Recent Medicare reimbursement changes are placing stricter limits on these high-cost therapies, creating an opening for new technologies to enter the market.
“We see this as an opportunity to introduce a first-line-of-care product for patients,” Krebs explained. “It’s a highly innovative technology entering a market that has seen little innovation in decades.”
Inside the Innovation
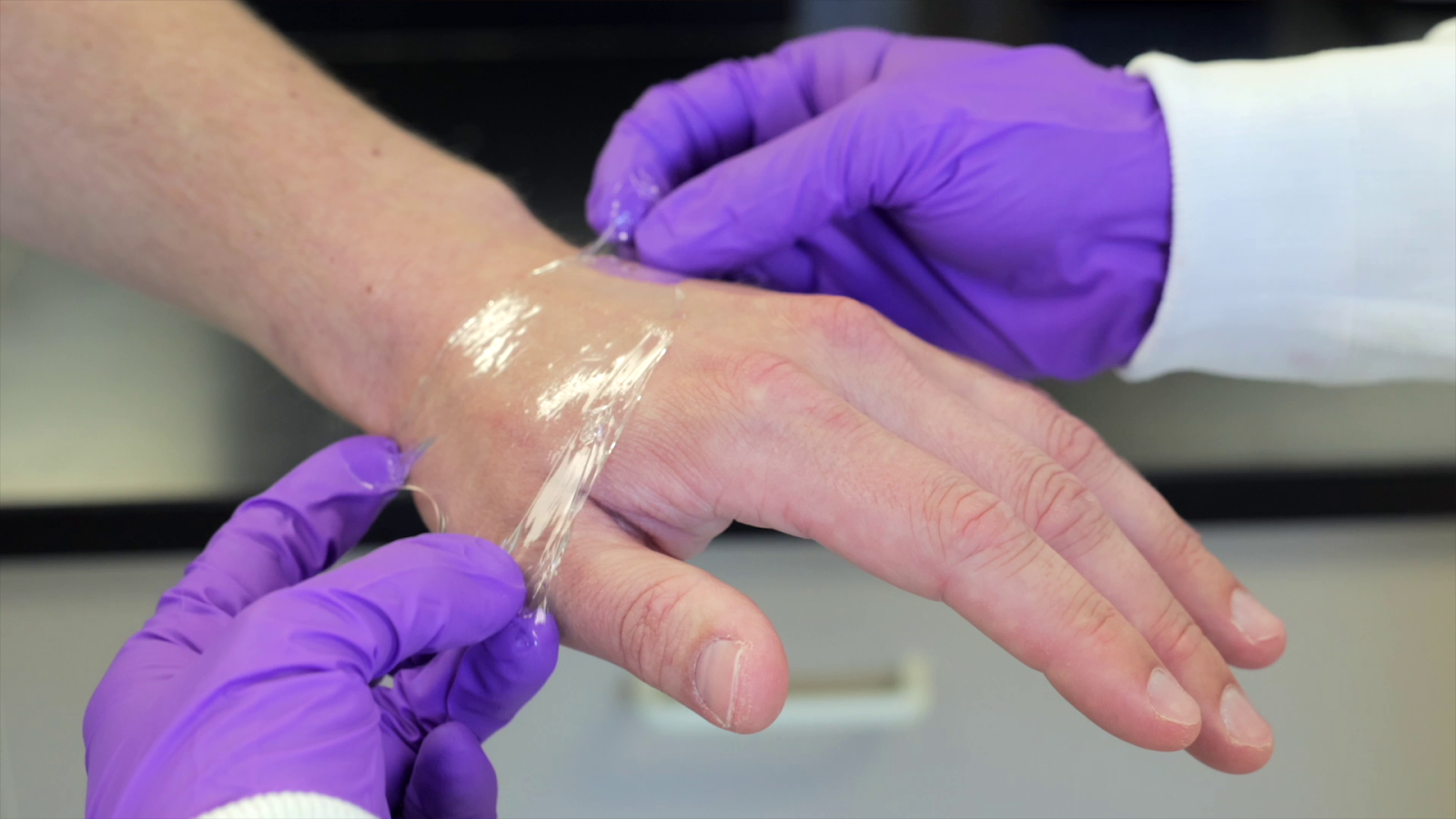
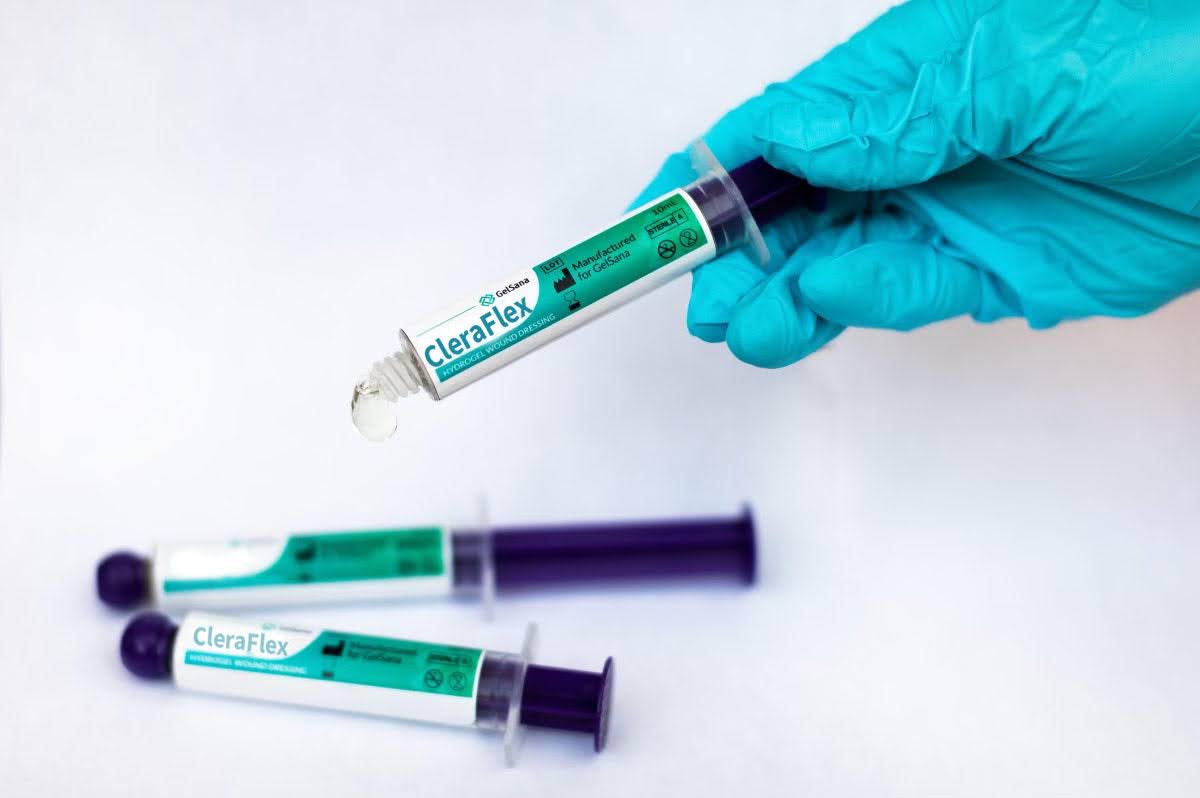
At the core of GelSana’s platform is a novel class of synthetic polymers based on zwitterionic chemistry.
“Our polymers are fully synthetic and built on what is known as zwitterionic chemistry,” Krebs explained. “The term refers to polymers that carry closely spaced positive and negative charges along the backbone, which gives the material a very unique set of properties.”
This molecular structure enables the material to behave very differently from conventional wound dressings.
“These materials are essentially invisible to the body and completely evade the foreign body response,” Krebs said. “They also repel bacteria, which makes them very promising from an infection prevention standpoint.”
Preclinical studies suggest the polymers can help reduce inflammation at the wound site, creating a more favorable environment for tissue repair.
“The really difficult-to-heal wounds are often highly inflamed and very prone to infection,” Krebs said. “We engineered these materials specifically for that environment, where helping to calm inflammation and preventing infection can stop wounds from escalating into more serious complications.”
GelSana’s first commercial product, CleraFlex Stretchable Wound Dressing, applies this polymer technology in a format designed for clinical usability. The dressing is extrudable and highly elastic, allowing clinicians to apply it precisely while it gently adheres to surrounding healthy tissue without sticking to the moist wound bed. As a partially hydrated material, it helps maintain moisture balance by both donating hydration to dry wounds and absorbing excess fluid from wounds with exudate.
The material can also serve in the future as a delivery platform for therapeutics, enabling controlled release of drugs directly into the wound bed to further accelerate healing.
Progress and Milestones
GelSana has made rapid progress since its founding in 2020. The company has completed extensive preclinical testing across multiple experimental models.
“We’ve completed extensive preclinical testing, including in vitro work as well as both small and large animal studies,” Krebs shared.
The company is now on the verge of bringing its first product to market. CleraFlex will be introduced as a Class I wound dressing device that is exempt from the 510(k) clearance process.
“Once it is registered with the FDA, clinicians can begin using it immediately, which allows us to move much faster toward commercialization,” she said.
GelSana has also participated in leading startup accelerator programs, including MedTech Innovator and Plug and Play. These programs connect emerging companies with major strategics and investors across the medtech ecosystem.
Looking ahead, GelSana is currently raising a $10 million Series A round to support manufacturing scale-up and commercial expansion.
“Our goal is to scale revenue and clinical adoption to the point where the company becomes an attractive acquisition target,” Krebs says. “But getting there requires building a strong business and sustainable revenue streams first.”
With a lean team, a strong scientific foundation, and a novel materials platform entering the clinic, GelSana is positioning itself to bring meaningful innovation to a wound care market that has seen limited material advances in recent decades.
Join Us at LSI USA ‘26
Krebs has been selected to present at LSI USA ‘26, March 16th–20th, in front of hundreds of global medical technology companies. Join us in welcoming her to the event in Dana Point, CA, where she will share the latest updates on GelSana’s technology and development.
17011 Beach Blvd, Suite 500 Huntington Beach, CA 92647
714-847-3540© 2026 Life Science Intelligence, Inc., All Rights Reserved. | Privacy Policy | Your Privacy Choices | Delete my Data